Kidney Function and GIST Drugs
 GIST Support International posed questions about the effects of drugs used to treat GIST on kidney function to Benjamin Humphreys, MD PhD. Dr. Humphreys is a nephrologist at Brigham and Women’s Hospital and Dana Farber Cancer Institute. In his clinical practice at the Dana Farber Cancer Institute, he specializes in kidney care for cancer patients. He co-directs the Harvard Stem Cell Institute Kidney Program, and in his basic science lab he investigates kidney injury and repair in order to translate this knowledge into therapies for patients with kidney disease. He is also pursuing translational studies of cancer patients who receive antiangiogenic therapies, focusing on the mechanisms of hypertension, proteinuria and kidney injury that these drugs can induce. Understanding these mechanisms may lead to new approaches to prevent kidney damage and new predictive biomarkers to measure tumor response may be discovered. Dr. Humphreys has published 25 peer-reviewed original publications and 24 review articles or book chapters, and he lectures regularly on kidney and blood pressure complications of cancer therapy.
GIST Support International posed questions about the effects of drugs used to treat GIST on kidney function to Benjamin Humphreys, MD PhD. Dr. Humphreys is a nephrologist at Brigham and Women’s Hospital and Dana Farber Cancer Institute. In his clinical practice at the Dana Farber Cancer Institute, he specializes in kidney care for cancer patients. He co-directs the Harvard Stem Cell Institute Kidney Program, and in his basic science lab he investigates kidney injury and repair in order to translate this knowledge into therapies for patients with kidney disease. He is also pursuing translational studies of cancer patients who receive antiangiogenic therapies, focusing on the mechanisms of hypertension, proteinuria and kidney injury that these drugs can induce. Understanding these mechanisms may lead to new approaches to prevent kidney damage and new predictive biomarkers to measure tumor response may be discovered. Dr. Humphreys has published 25 peer-reviewed original publications and 24 review articles or book chapters, and he lectures regularly on kidney and blood pressure complications of cancer therapy.
Below are Dr. Humphrey’s expert responses to our questions.
1) Explain briefly the structure and function of the kidney, particularly the renal glomerulus filtration unit.
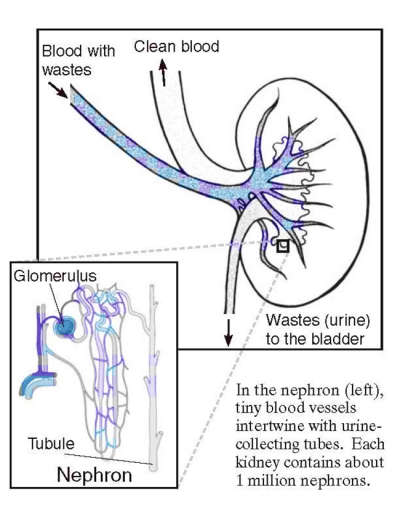 |
| Schematic illustration of the nephron unit within the kidney (source: NIDDK, http://www2.niddk.nih.gov/). |
The kidney has two main functions:
- It filters the blood to regulate fluids, electrolytes and blood pressure, and
- it acts as an endocrine organ to regulate red blood cell production and bone health.
The functional unit of the kidney is the nephron, and humans have about one million nephrons in each of their kidneys. The head of the nephron is the glomerulus – this structure is shaped like a ball, and blood enters it on one side where it is filtered to create what will become the urine. However, cells and larger proteins are kept inside the blood stream – they are kept out of the urine. This filtrate then travels through a long tube, the body of the nephron, where water and electrolytes are reabsorbed into the bloodstream except for a small fraction which ultimately leaves the nephron at the other end, traveling into the collecting duct, the ureter and then the bladder. Toxins and drugs are not reabsorbed, however, and end up in the urine.
To illustrate the reabsorption that goes on after the fitrate is formed at the glomerulus, about 180 Liters of filtrate are formed in a person each day, but only about 1.5 Liters of urine are ultimately excreted. So, urine represents less than 1% of what is actually filtered at the glomerulus.
2) Describe the kidney toxicities that occur with targeted cancer therapies.
Targeted cancer therapies have had a tremendous positive impact on the treatment of many cancers, and GIST of course is perhaps the best example. Despite their effectiveness, initial hopes that this class of therapy would have few or no toxicities have given way to an appreciation that there are unique side effects of this drug class. In kidney, the two most common types of toxicity are:
- hypertension (which reflects the combined effects of kidney, blood vessels and heart), and
- proteinuria, or leakage of protein into the urine.
3) Briefly explain the molecular mechanisms of these toxicities.
The mechanisms of anti-VEGF therapy-induced hypertension are not fully understood, but the last three years have seen important advances in our understanding of some of the pathophysiology. VEGF secretion by cancers is one way the tumor attracts new blood vessel growth (the process of tumor-angiogenesis), so it can receive the nutrients it needs to grow. But it turns out that VEGF also promotes relaxation of normal blood vessels, especially in the microcirculation of the rest of the body. When VEGF signaling is blocked by targeted therapies, the relaxing effect is blocked, causing systemic vasoconstriction. This narrowing of the caliber of the microcirculation forces the heart to pump harder to deliver the same amount of blood to the organs, raising blood pressure. It is likely that anti-VEGF drugs also impact the kidney’s ability to regulate sodium balance, which ultimately is the primary regulator of blood pressure. This is an important future area of research.
How targeted therapies cause proteinuria is less clear. The glomerulus is the filtering unit in the kidney, normally allowing fluids through but not proteins. Ultimately the fluid filtered by the glomerulus becomes urine. The central cell type in the glomerular filter is the podocyte – a beautiful cell that forms a very delicate, selective barrier to the blood. Podocytes also happen to be among the most highly secreting cells of VEGF in the entire body. Podocyte-derived VEGF acts on nearby endothelial cells to help keep the barrier function of the glomerulus intact. Anti-VEGF therapies disrupt this signaling, though the precise mechanism is unknown. Once damaged, the podocyte filter becomes leaky, allowing protein in the urine. [See the electron micrographs below illustrating the structures that accomplish kidney filtration; these micrographs are from the NIDDK website.]
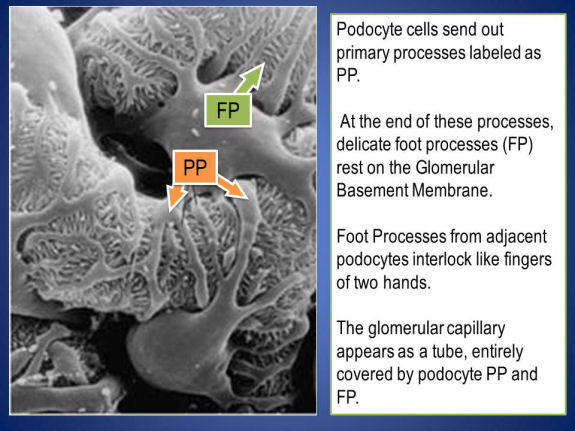
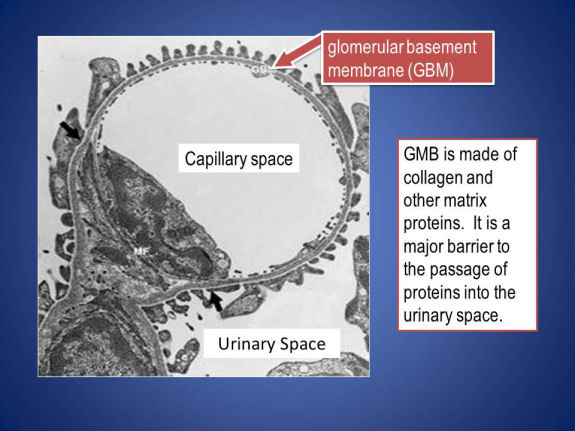
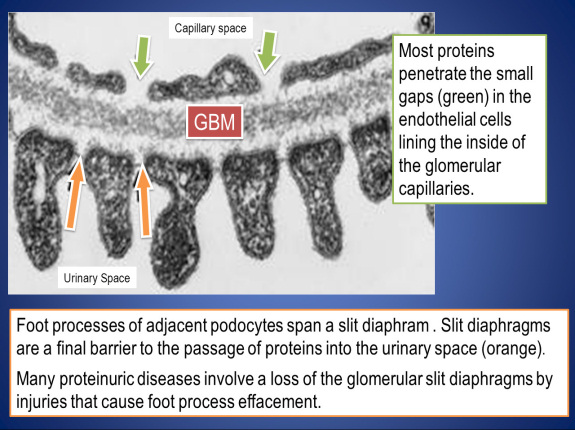
A particularly interesting aspect to these toxicities is that in some cases, it appears that patients who develop hypertension may have a superior cancer response to the targeted therapy. This is because unlike traditional chemotherapy, where toxicity usually represented an “off-target” effect that was unanticipated, toxicities from targeted therapies represent “on-target” effects, that is, consequences of blocking the signaling pathways that they were intended to block. So patients who develop hypertension do so because their VEGF pathway has been effectively blocked. This gives confidence that their tumor-VEGF pathway is similarly being blocked, providing an explanation for why patients that develop hypertension have superior outcomes in some cases. This idea is now being tested prospectively, as investigators are now assessing whether increasing the dose of anti-VEGF therapies in patients that do not develop hypertension improves their cancer outcome.
There are many unanswered questions in this area: Might proteinuria also correlate with outcome, since it is also an “on-target” toxicity? What about the non-VEGF pathways that many antiangiogenics inhibit as well – such as the PDGF pathway in the case of Imatinib – might these account for some of the toxicities observed? How should these toxicities be best managed to optimize the anticancer benefit while minimizing the very real morbidities that can be caused by uncontrolled hypertension or proteinuria? Will additional toxicities become clear as patients live longer on treatment?
4) What symptoms might patient
s notice who experience these toxicites? What clinical tests are performed to monitor kidney function?
Hypertension is generally asymptomatic – the reason that regular monitoring is important. With extremely high blood pressure, patients may develop headache, visual disturbances, chest pain or shortness of breath.
The most common clinical test used to measure kidney function is serum creatinine. This is a very common test that most patients will have checked on all regular lab draws. When the serum creatinine rises, it indicates that the kidney is not working as well as it should, because the kidney usually excretes creatinine efficiently. So, a buildup of creatinine (usually) means that the kidney is not filtering creatinine, and everything else, as it should.
Another common clinical test that reflects kidney function is urinalysis. This can indicate whether the kidney is leaking protein or blood cells. Either of these would indicate a potential kidney problem, though they can also be abnormal for other reasons such as urinary tract infection, or a kidney stone.
5) How does kidney dysfunction affect the body in the short term and long term?
In the short term, kidney injury can lead to the buildup of toxins and fluid, causing swelling, high blood pressure and laboratory abnormalities. If the kidney is injured very severely, it can shut down entirely and stop producing urine. In this case dialysis might be required. Fortunately, the kidney has a very strong capacity to repair itself, and it often recovers and patients come off of dialysis when their kidneys start functioning again.
Over the long term, the slow loss of kidney function can lead to hypertension, swelling, anemia, and disorders of bone metabolism. There is also a strong association between chronic kidney disease and heart disease. It can also hamper the body’s ability to excrete drugs – making it more difficult to safely administer certain therapies, for example.
6) What actions should be taken if drug-induced kidney dysfunction is noted in a GIST patient?
This will depend on what the dysfunction is. In the case of hypertension, this is almost always treatable with antihypertensive medications. With the development of proteinuria, usually patients can still continue on therapy but they require monitoring of the amount of protein in the urine, and potentially new medications that reduce the level of proteinuria. Most of the time these complications can be managed by the oncologist, but sometimes consultation with a nephrologist can be helpful. A sustained rise in the serum creatinine should usually be evaluated by a nephrologist as well.
There are several things that a patient with chronic kidney disease (CKD, the term for permanent damage, as reflected by sustained elevation in serum creatinine) can do to help avoid further kidney damage. They should avoid iodinated contrast when possible (the dye that can be given with CT scans),
because this can lead to an acute worsening of kidney function. Patients that have a normal creatinine can undergo CT scanning with contrast dye safely. Patients with GIST and CKD should avoid non-steroidal anti-inflammatory drugs (Ibuprofen, Alleve, etc…) which sometimes can be hard for GIST patients, since some therapies such as Gleevec cause aches and pains as a side effect. But these over the counter drugs can also harm the kidney if taken regularly (every once in a while is fine). Attention to blood pressure control is important, and a low salt diet can help, but usually antihypertensive medications are required. The National Kidney Foundation has a helpful, patient-oriented site that
addresses many of these issues in much more detail.

